Spectroscopy


In order to be able to study and analyse light, one can use a spectroscope to view the component wavelengths of a source.
A spectroscope is a simple device that uses a dispersive element such as a prism or a diffraction grating to separate out the spectral components. The input of the spectroscope contains the slit, which is an important component in the system, as this determines the spectral resolution of the system.
The dispersive element will determine the bandwidth of the instrument.


Paton Hawksley in the UK manufacture some high-quality Spectroscopes, (my ones pictured top left) and can I say these are very well engineered products. Two versions of the spectroscope are available, both feature a 600 line/mm transmission diffraction grating.
The larger benchtop spectroscope is the TE313, while its smaller sibling is the TE310 pocket spectroscope. For further
details and to purchase these spectroscopes, head over to:
Paton Hawksley Education Limited.
The image to the left is an iPhone photo taken (with difficulty) through the eye piece of the TE313 Benchtop Spectroscope. However, the actual optical resolution of this Spectroscope is excellent and produces very sharp lines when viewed directly. At some stage will see if possible, to setup an ability to obtain a high-quality photo of the Spectrum produced by this spectroscope.
My only desire would a calibrated wavelength reference viewable within the field of view of the spectroscope.
A spectrometer is a device that takes an input signal (in this case, light) and separates the signal into its into its component wavelengths. A CCD imaging detector converts the light signal into an electrical signal and processed by the electronics into a digital data value.
The spectrometer is typically connected to a computer that is running the acquisition and analysis software and presents the data as a spectrograph.
In the next section I will discuss a surplus Spectrometer module that is manufactured by B&WTek in the USA, and have been fortunate to have been able to obtain 3 of them from Ebay back in 2018.
The BTC110-S spectrometer module is a custom module that was used in a Bio-photonic carotenoid antioxidants scanner. The carotenoid scanner uses Resonant Raman Spectroscopy to determine the level of antioxidants in a patient.
The Raman effect (and Raman Spectroscopy) is a process where the excitation wavelength (in this case 473nm laser) interacts with matter where light energy is transferred to molecular vibrations, (in this case, the Carotenoid molecule).
The scattered light is detected using the scanner's spectrometer that has been setup to detect the scattered wavelength. In the case of the carotenoid molecule, this shifts the 473nm photo to around 518nm in the green.
The configuration of the spectrometer's optical bench is the crossed Czerny-Turner configuration. The BTC110-S utilises the Sony ILX511 CCD linear image sensor which features a 14um pixel size.
For detailed article on how the spectrometer works and the various configurations, head on over to the B&W Tek website: How Does a Spectrometer Work?.

B&WTek BTC110-S Spectrometer
To make use of the spectrometer module, some modification (and if needed, realignment of the optical bench) are required.
The spectrometer has two internal baffles that limit the bandwidth, as well as a notch filter at the slit to further filter out the excitation laser wavelength that can mask the very weak Raman scattered signal.
Following the basic modifications, the spectrometer's useful bandwidth is around 200nm with the default installed 1800 line/mm grating, and capable of up to 1nm resolution with a very well aligned optical bench.
By comparison, a 900 line/mm grating would gain a bandwidth of around 545nm at a resolution of 2nm and a 600 line/mm grating, a bandwidth of around 735nm and a resolution of around 3nm.
Following the modifications (and alignment) of the optical bench, the spectrometer must be calibrated.
The calibration process requires the use of a spectral light source and or lasers as they contain specific known wavelength peaks.
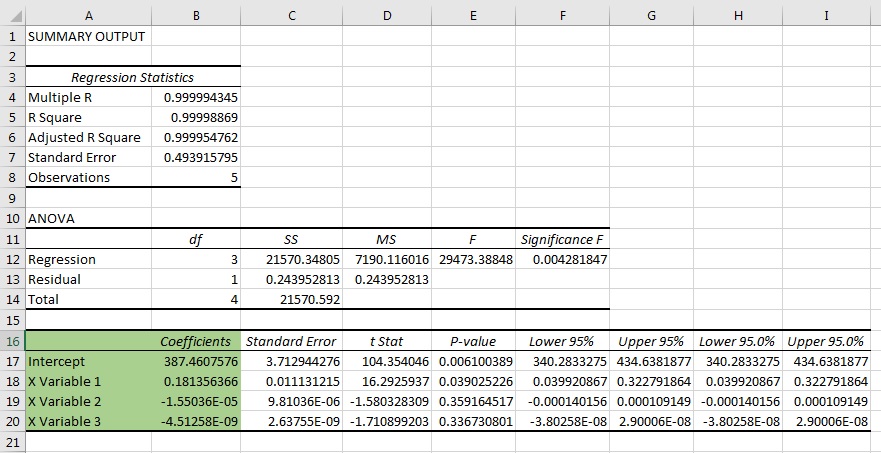
Within the software, the spectrometer's coefficient values of C0, C1 , C2 and C3 are reset to C0=0, C1=1, C2=0 and C3=0. In this configuration, the X-Axis of the graph is referenced as pixel numbers from Pixel 0 to Pixel 2047.
Each of the predominant wavelength peaks are identified against the pixel number they fall on, and the values are noted down.
The next step, the wavelength vs pixel values are fitted to a third-order polynomial equation. . .
λp = I + C1P + C2P2 + C3P3
. . . Where, λ is the wavelength at pixel number, p, I, is the wavelength of pixel 0, C1 the first coefficient (nm/pixel), C2 the second coefficient (nm/pixel2), and C3 the thid coefficient (nm/pixel3).
The output of the equation outputs the coefficient values to calibrate the wavelength scale.
These coefficient values can be calculated using Excel's linear regression, which is found in the add-on data analysis tools. From the output of tool, the values used are the Intercept, X Variable 1, X Variable 2, X Variable 3 (green shaded area).
Read more about the application of this spectrometer to measure the wavelengths on my Dye Laser project study.
The following Image is a Fine Art edition print of the spectrum of light. The Spectroscope used to be able to create this image was a home setup using a diffraction grating from an old Pioneer Laser Disk Player assembly and my home-made slits.
Recently, I have procured a small collection of low pressure sodium (LPS) lamps and LPS spectral lamps for some experimentation of the unique, monochromatic light that they produce.
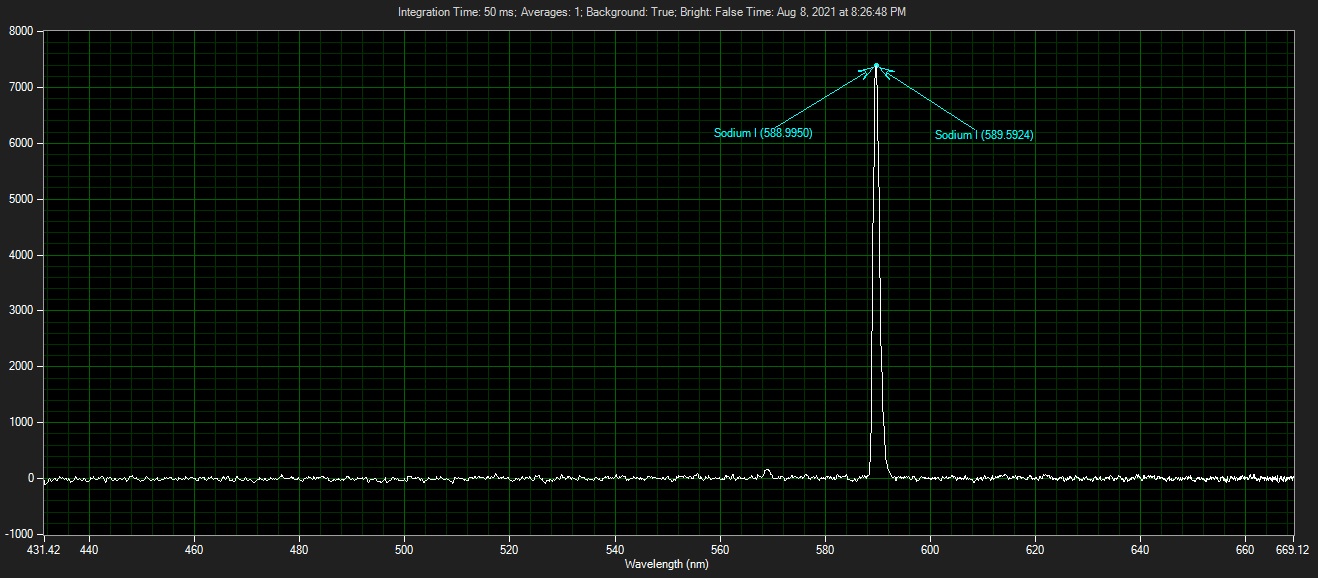
Low pressure sodium lamps produce a characteristic yellow light with a wavelength of 589nm, strictly, two very close wavelengths at 589nm and 589.6nm known as the sodium doublet or "D-Lines".
The above spectrograph produced using the B&W Tech Spectrometer.
With the installed 1800 line/mm grating, the best resolution available for a well aligned spectrometer is 1nm. The doublet lines of the Sodium lamp is only 0.6nm separation, i.e. would need a far better spectrometer or interferometer to be able to visualise the lines.
One facinating experiment that can be demonstrated using a low pressure sodium lamp is that of the Black flame experiment.
- Flavio Spedalieri -
Written: 26th July 2021
Updated: 9th August 2021
This Document Complies to W3C
XHTML 1.0 Strict Standards